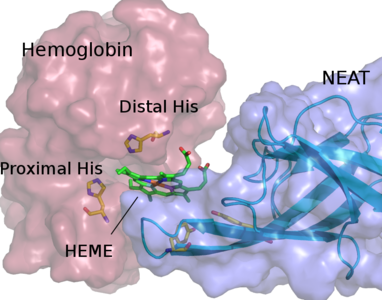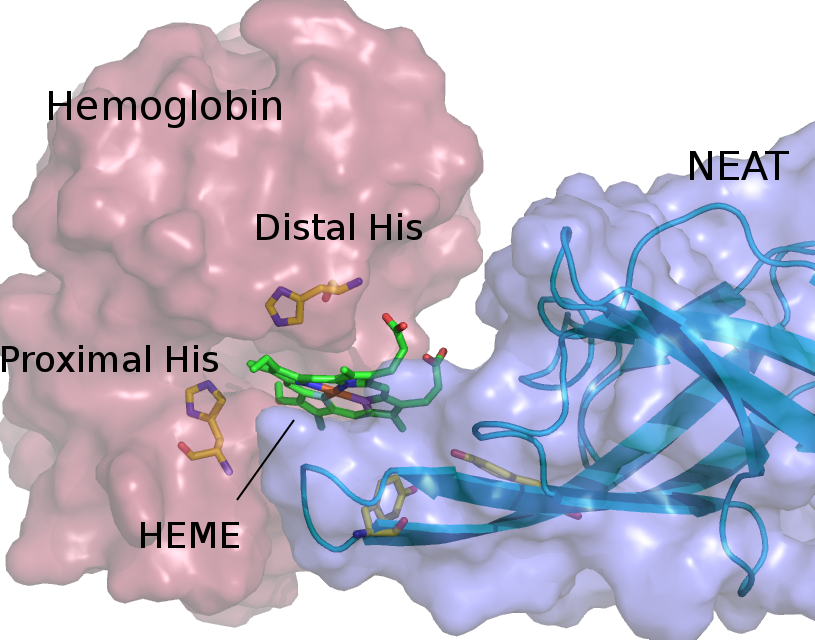
Red blood cell hemolysis in sickle cell disease (SCD) releases free hemoglobin. Extracellular hemoglobin and its degradation products, free heme and iron, are highly toxic due to oxidative stress induction and decrease in nitric oxide availability. We propose an approach that helps to eliminate extracellular hemoglobin toxicity in SCD by employing a bacterial protein system that evolved to extract heme from extracellular hemoglobin. NEAr heme Transporter (NEAT) domains from iron-regulated surface determinant proteins from Staphylococcus aureus specifically bind free heme as well as facilitate its extraction from hemoglobin. We demonstrate that a purified NEAT domain fused with human haptoglobin β-chain is able to remove heme from hemoglobin and reduce heme content and peroxidase activity of hemoglobin. We further use molecular dynamics (MD) simulations to resolve molecular pathway of heme transfer from hemoglobin to NEAT, and to elucidate molecular mechanism of such heme transferring process. Our study is the first of its kind, in which simulations are employed to characterize the process of heme leaving hemoglobin and subsequent rebinding with a NEAT domain. Our MD results highlight important amino acid residues that facilitate heme transfer and will guide further studies for the selection of best NEAT candidate to attenuate free hemoglobin toxicity.
Publications:
Sakipov S, Rafikova O, Kurnikova MG, Rafikov R. Molecular mechanisms of bio-catalysis of heme extraction from hemoglobin Redox Biol. 11:516-523. (2017)